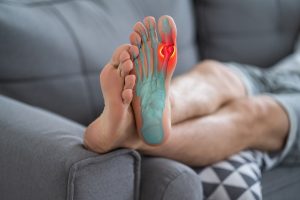
The Cartiva toe implant has become a commonly used device aimed at relieving symptoms of arthritis in the big toe. It has, however, been known to fail at an abnormally high rate, causing pain, loss of motion, and even additional surgery. If you or someone you know has experienced adverse health effects from receiving the Cartiva toe implant, you may want to keep reading.
About the Cartiva Implant
The Cartiva toe implant was manufactured to treat symptoms of big toe arthritis. This type of arthritis occurs when cartilage is damaged, or worn down, in the big toe joint. The degraded cartilage causes the bones to rub together, which can lead to pain, stiffness, and swelling. The Cartiva implant is a synthetic replacement for the degraded cartilage. Its primary purpose is to reduce bone-on-bone friction in the metatarsophalangeal joint in the big toe. It is specifically molded from Polyvinyl Hydrogel (PVA Hydrogel), and is roughly the size of a popcorn kernel. Prior to this implant, the only viable treatment option for big toe arthritis was a difficult fusion surgery that left the patient with limited mobility. This product became the first openly accessible alternative to fusion surgery.
 North Carolina Product Liability Lawyer Blog
North Carolina Product Liability Lawyer Blog

